Inflammatory Bowel Disease

Measure & monitor mucosal healing changes over time
Improvement in mucosal damage provides an objective measure of therapeutic response.1,2 However, current assessment methods may not provide needed information, particularly when evaluating disease activity in difficult-to-reach locations to assess ideal disease.1-4
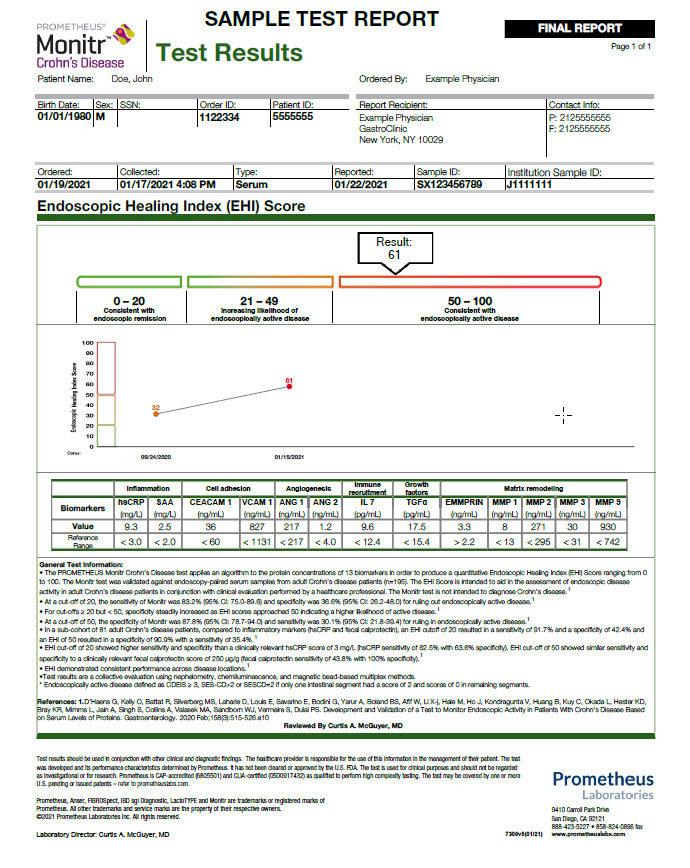
Monitr® is a laboratory-developed test that evaluates multiple markers of mucosal damage and repair processes, regardless of disease location.5 It applies a proprietary algorithm to 13 biomarkers to produce a quantitative Endoscopic Healing Index (EHI) Score—ranging from 0 to 100—to aid in distinguishing endoscopic remission from active disease in IBD patients.


The next evolution
of the treat-to-target paradigm
In IBD patient management, endoscopic healing is known to be an objective measure of therapeutic response.4 In multiple clinical trials, endoscopic healing is a therapeutic goal associated with improved long-term outcomes4-7.
However, current assessment methods of disease activity can make it challenging to:
- Noninvasively evaluate the endoscopic disease activity 2,8,9
- Assess active disease in difficult-to-reach locations, including the ileum4,7,9
- Obtain objective results for individual patients2,7
- Regularly monitor patient response to therapy2,7
Measure and monitor the endoscopic disease activity with one simple serum test
The Monitr Crohn’s Disease Test is a laboratory-developed test that provides critical information to aid in:
- Establishing a baseline measurement of disease activity at initial presentation
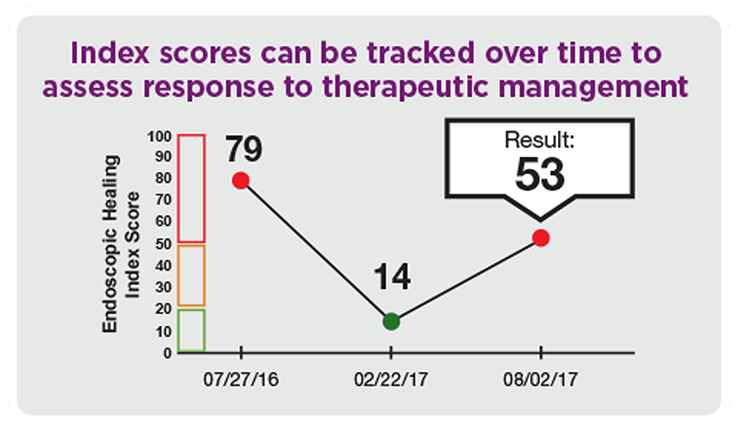
- Tracking and assessing disease activity and response to therapeutic management
- Optimizing treatment management decisions
- Evaluating if treatment changes may be appropriate
- Counseling patients about therapeutic management goals
References
- Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015;110(9):1324-1338.
- Alsoud D, Ho J, Sabino J, Ferrante M, Vermeire S, Verstockt B. Utility of the Serum-Based Endoscopic Healing Index in Monitoring Therapeutic Response in Ulcerative Colitis. Am J Gastroenterol. 2023 Oct 19. doi: 10.14309/ajg.0000000000002518.
- Colombel JF, Panaccione R, Bossuyt P, et al. Effect of tight control management on Crohn’s disease (CALM): a multicentre, randomised, controlled phase 3 trial. Lancet. 2017;390(10114):2779-2789
- De Cruz P, Kamm MA, Prideaux L, Allen PB, Moore G. Mucosal healing in Crohn’s disease: a systematic review. Inflamm Bowel Dis. 2013;19(2):429-444.
- D’Haens G, Kelly O, Battat R, et al. Development and validation of a test to monitor endoscopic activity in patients with Crohn’s disease based on serum levels of proteins. Gastroenterology. 2020;158(3):515-526.e10.
- Rutgeerts P, Van Assche G, Sandborn WJ, et al. Adalimumab induces and maintains mucosal healing in patients with Crohn’s disease: data from the EXTEND trial. Gastroenterology. 2012;142(5):1102-1111.e2.
- Shah SC, Colombel JF, Sands BE, Narula N. Systematic review with meta-analysis: mucosal healing is associated with improved long-term outcomes in Crohn’s disease. Aliment Pharmacol Ther. 2016;43(3):317-333.
- Freeman HJ. Limitations in assessment of mucosal healing in inflammatory bowel disease. World J Gastroenterol. 2010;16(1):15-20.
- Bryant RV, Winer S, Travis SP, Riddell RH. Systematic review: histological remission in inflammatory bowel disease. Is ‘complete’ remission the new treatment paradigm? An IOIBD initiative. J Crohns Colitis. 2014;8(12):1582-1597.